 Similarly, the elements of Group 17 have a similar outer electronic configuration, ns 2np 5.Like, for instance, all the elements of Group 1 have a similar outer electronic configuration, ns 1, where n refers to the Principal Quantum Number of the outermost shell.The repetition of the similar outer electronic configuration after certain regular intervals is the cause of periodicity in properties. What is the Cause of Periodicity in the Modern Periodic Table? Each group in the periodic table consists of elements that have the same outer shell electronic configuration.There are 18 groups in the long form of the periodic table that are numbered from 1 to 18.The atomic radii of the elements increase from the start to finish in a group. The elements in a similar group generally show patterns in atomic radii, ionization energy, and electronegativity. The vertical segments, i.e, the columns in the modern or long form of the periodic table are known as Groups. Apart from that, there is a separate panel at the bottom of the long form of the periodic table that consists of 14 elements from the 6th period called the lanthanoids and 14 elements from the 7th period called the actinoids. 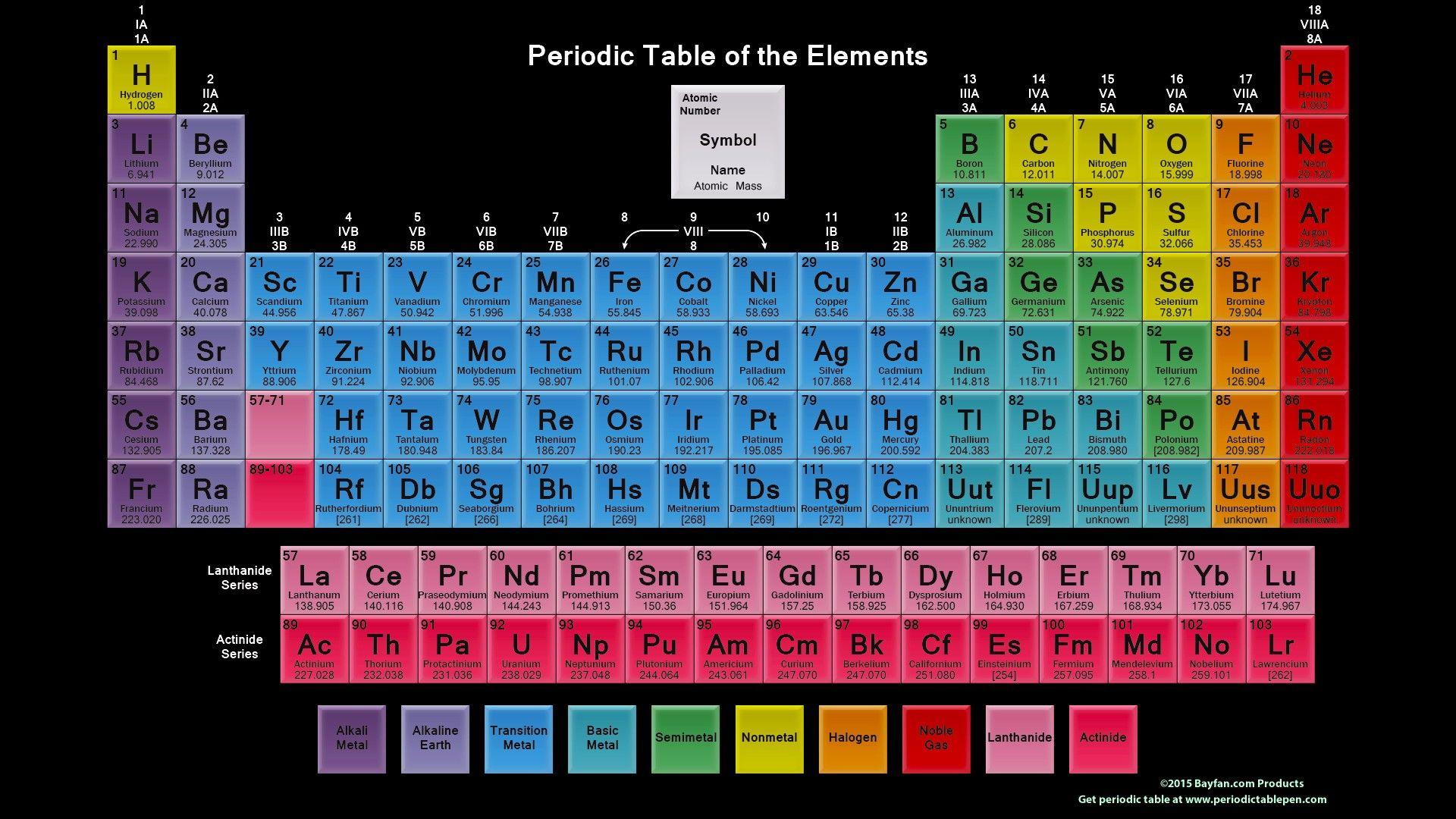
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
